Mercallite
A valid IMA mineral species - grandfathered
This page is currently not sponsored. Click here to sponsor this page.
About Mercallite
Formula:
KHSO4
Colour:
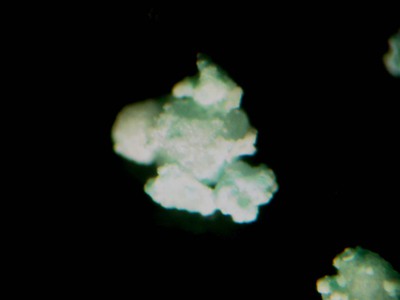
Colourless, sky-blue; colourless in transmitted light
Lustre:
Vitreous
Specific Gravity:
2.31
Crystal System:
Orthorhombic
Name:
Named in 1935 by Guido Carobbi in honor of Giuseppe Mercalli (21 May 1850, Milan, Italy – 19 March 1914, Naples, Italy), volcanologist. At different times he was professor of geology at the University of Catania, professor at Naples University, and Director of the Vesuvius Observatory.
This page provides mineralogical data about Mercallite.
Unique Identifiers
Mindat ID:
2646
Long-form identifier:
mindat:1:1:2646:4
GUID
(UUID V4):
(UUID V4):
267128f4-c551-457a-a6ae-7e38b73d3e21
IMA Classification of Mercallite
Approved, 'Grandfathered' (first described prior to 1959)
IMA Formula:
KH(SO4)
Classification of Mercallite
7.AD.10
7 : SULFATES (selenates, tellurates, chromates, molybdates, wolframates)
A : Sulfates (selenates, etc.) without additional anions, without H2O
D : With only large cations
7 : SULFATES (selenates, tellurates, chromates, molybdates, wolframates)
A : Sulfates (selenates, etc.) without additional anions, without H2O
D : With only large cations
28.1.1.1
28 : ANHYDROUS ACID AND NORMAL SULFATES
1 : Anhydrous Acid Sulfates
28 : ANHYDROUS ACID AND NORMAL SULFATES
1 : Anhydrous Acid Sulfates
25.1.5
25 : Sulphates
1 : Sulphates of the alkali metals and ammonium
25 : Sulphates
1 : Sulphates of the alkali metals and ammonium
Mineral Symbols
As of 2021 there are now IMA–CNMNC approved mineral symbols (abbreviations) for each mineral species, useful for tables and diagrams.
| Symbol | Source | Reference |
|---|---|---|
| Mec | IMA–CNMNC | Warr, L.N. (2021). IMA–CNMNC approved mineral symbols. Mineralogical Magazine, 85(3), 291-320. doi:10.1180/mgm.2021.43 |
Physical Properties of Mercallite
Vitreous
Transparency:
Transparent
Colour:
Colourless, sky-blue; colourless in transmitted light
Cleavage:
None Observed
Density:
2.31 g/cm3 (Measured) 2.32 g/cm3 (Calculated)
Optical Data of Mercallite
Type:
Biaxial (+)
RI values:
nα = 1.445 nβ = 1.460 nγ = 1.491
2V:
Measured: 56° , Calculated: 72°
Max Birefringence:
δ = 0.046

Image shows birefringence interference colour range (at 30µm thickness)
and does not take into account mineral colouration.
and does not take into account mineral colouration.
Surface Relief:
Moderate
Dispersion:
relatively strong
Chemistry of Mercallite
Mindat Formula:
KHSO4
Elements listed:
Crystallography of Mercallite
Crystal System:
Orthorhombic
Class (H-M):
mmm (2/m 2/m 2/m) - Dipyramidal
Space Group:
Pbca
Setting:
Pbca
Cell Parameters:
a = 8.429(3) Å, b = 18.976(3) Å, c = 9.807(3) Å
Ratio:
a:b:c = 0.444 : 1 : 0.517
Unit Cell V:
1,568.62 ų (Calculated from Unit Cell)
Z:
16
Morphology:
Stalactites composed of minute tabular crystals. Crystals tabular {001} and elongated [100] at times (artificial).
Crystal Structure
Load
Unit Cell | Unit Cell Packed
2x2x2 | 3x3x3 | 4x4x4
Unit Cell | Unit Cell Packed
2x2x2 | 3x3x3 | 4x4x4
Show
Big Balls | Small Balls | Just Balls | Spacefill
Polyhedra Off | Si Polyhedra | All Polyhedra
Remove metal-metal sticks
Big Balls | Small Balls | Just Balls | Spacefill
Polyhedra Off | Si Polyhedra | All Polyhedra
Remove metal-metal sticks
Display Options
Black Background | White Background
Perspective On | Perspective Off
2D | Stereo | Red-Blue | Red-Cyan
Black Background | White Background
Perspective On | Perspective Off
2D | Stereo | Red-Blue | Red-Cyan
View
CIF File Best | x | y | z | a | b | c
CIF File Best | x | y | z | a | b | c
Rotation
Stop | Start
Stop | Start
Labels
Console Off | On | Grey | Yellow
Console Off | On | Grey | Yellow
Data courtesy of the American Mineralogist Crystal Structure Database. Click on an AMCSD ID to view structure
| ID | Species | Reference | Link | Year | Locality | Pressure (GPa) | Temp (K) |
|---|---|---|---|---|---|---|---|
| 0009207 | Mercallite | Loopstra L H, MacGillavry V H (1958) The crystal structure of KHSO4 (mercallite) Acta Crystallographica 11 349-354 |  | 1958 | synthetic | 0 | 293 |
| 0009528 | Mercallite | Cotton F A, Frenz B A, Hunter D L (1975) The structure of potassium hydrogensulfate Acta Crystallographica B31 302-304 |  | 1975 | synthetic | 0 | 293 |
| 0009570 | Mercallite | Payan F, Haser R (1976) On the hydrogen bonding in potassium hydrogen sulphate. Comparison with a previous crystal structure determination Acta Crystallographica B32 1875-1879 |  | 1976 | synthetic | 0 | 293 |
CIF Raw Data - click here to close
X-Ray Powder Diffraction
Powder Diffraction Data:
| d-spacing | Intensity |
|---|---|
| 3.84 Å | (100) |
| 3.52 Å | (85) |
| 3.41 Å | (85) |
| 3.26 Å | (85) |
| 3.87 Å | (65) |
| 3.03 Å | (65) |
| 2.472 Å | (30) |
Geological Environment
Paragenetic Mode(s):
| Paragenetic Mode | Earliest Age (Ga) |
|---|---|
| Stage 7: Great Oxidation Event | <2.4 |
| 45a : [Sulfates, arsenates, selenates, antimonates] | |
| 47a : [Near-surface hydration of prior minerals] | |
| 47b : [Sulfates and sulfites] |
Type Occurrence of Mercallite
Place of Conservation of Type Material:
University of Florence, Florence, Italy, 1976/I.
Geological Setting of Type Material:
Active volcanic fumaroles.
Associated Minerals at Type Locality:
Reference:
Carobbi G (1935) «Mercallite» nuovo minerale fra i prodotti dell'attivita fumarolica vesuviana del 1933. Rendiconti, Reale Accademia dei Lincei 21, 385-393
Other Language Names for Mercallite
Related Minerals - Strunz-mindat Grouping
| 7.AD. | Calciolangbeinite | K2Ca2(SO4)3 |
| 7.AD. | Bubnovaite | K2Na8Ca(SO4)6 |
| 7.AD. | Dobrovolskyite | Na4Ca(SO4)3 |
| 7.AD. | Murphyite | Pb(Te6+O4) |
| 7.AD. | Cuprodobrovolskyite | Na4Cu(SO4)3 |
| 7.AD. | Kristjánite | KNa2H(SO4)2 |
| 7.AD.05 | Arcanite | K2SO4 |
| 7.AD.05 | Mascagnite | (NH4)2SO4 |
| 7.AD.15 | Misenite | K8H6(SO4)7 |
| 7.AD.20 | Letovicite | (NH4)3H(SO4)2 |
| 7.AD.25 | Glauberite | Na2Ca(SO4)2 |
| 7.AD.25 | Thénardite | Na2SO4 |
| 7.AD.30 | Anhydrite | CaSO4 |
| 7.AD.30 | Metathénardite | Na2SO4 |
| 7.AD.35 | Anglesite | PbSO4 |
| 7.AD.35 | Baryte | BaSO4 |
| 7.AD.35 | Celestine | SrSO4 |
| 7.AD.35 | Olsacherite | Pb2(Se6+O4)(SO4) |
| 7.AD.40 | Kalistrontite | K2Sr(SO4)2 |
| 7.AD.40 | Palmierite | K2Pb(SO4)2 |
| 7.AD.45 | Ivsite | Na3H(SO4)2 |
| 7.AD.55 | Markhininite | TlBi(SO4)2 |
Other Information
Notes:
Soluble in water, yielding an acidic solution.
Health Risks:
No information on health risks for this material has been entered into the database. You should always treat mineral specimens with care.
Internet Links for Mercallite
mindat.org URL:
https://www.mindat.org/min-2646.html
Please feel free to link to this page.
Please feel free to link to this page.
Search Engines:
External Links:
Mineral Dealers:
References for Mercallite
Reference List:
Gossner, B. (1904) Zeitschrift für Kristallographie, Mineralogie und Petrographie, Leipzig: 39: 381.
Loopstra, L. H., MacGillavry, C. H. (1958) The crystal structure of KHSO4 (mercallite) Acta Crystallographica, 11 (5) 349-354 doi:10.1107/s0365110x58000943
Cruickshank, D. W. J. (1964) Refinements of structures containing bonds between Si, P, S or Cl and O or N. VIII. KHSO4 (mercallite) Acta Crystallographica, 17 (6) 682-683 doi:10.1107/s0365110x64001700
Cotton, F. A., Frenz, B. A., Hunter, D. L. (1975) The structure of potassium hydrogen sulfate. Acta Crystallographica Section B Structural Crystallography and Crystal Chemistry, 31 (1) 302-304 doi:10.1107/s0567740875002580
Localities for Mercallite
Locality List
 - This locality has map coordinates listed.
- This locality has map coordinates listed.
 - This locality has estimated coordinates.
ⓘ - Click for references and further information on this occurrence.
? - Indicates mineral may be doubtful at this locality.
- This locality has estimated coordinates.
ⓘ - Click for references and further information on this occurrence.
? - Indicates mineral may be doubtful at this locality.
 - Good crystals or important locality for species.
- Good crystals or important locality for species.
 - World class for species or very significant.
(TL) - Type Locality for a valid mineral species.
(FRL) - First Recorded Locality for everything else (eg varieties).
- World class for species or very significant.
(TL) - Type Locality for a valid mineral species.
(FRL) - First Recorded Locality for everything else (eg varieties).
All localities listed without proper references should be considered as questionable.
Canada | |
| Shang (2000) |
Costa Rica | |
| Ulloa et al. (2018) |
Iceland | |
| Balić-Žunić et al. (2016) |
Italy (TL) | |
| Accad. Lincei. Rend. Cl. Sci. fis. mat. nat. Roma (1935) +2 other references |
| Russo et al. (2004) | |
Russia | |
| Zelenski et al. (2020) |
Quick NavTopAbout MercalliteUnique IdentifiersIMA Classification Classification Mineral SymbolsPhysical Properties Optical Data Chemistry Crystallography Crystal StructureX-Ray Powder DiffractionGeological EnvironmentType Occurrence Other LanguagesStrunz-MindatOther InformationInternet Links References Localities Locality List





 symbol to view information about a locality.
The
symbol to view information about a locality.
The 



Mount Vesuvius, Somma-Vesuvius Complex, Naples, Campania, Italy